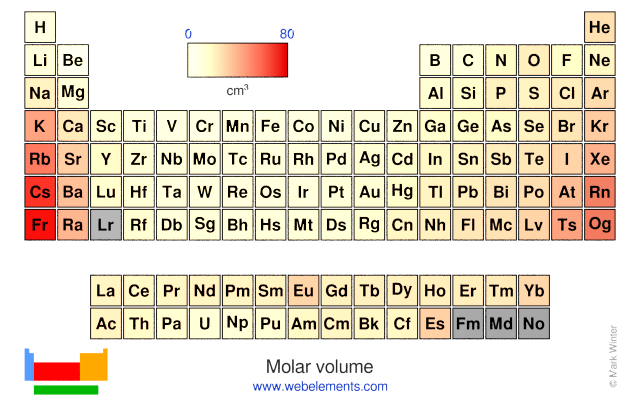
There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The unity for atomic mass is gram per mol. The lightest chemical element is Hydrogen and the heaviest is Hassium. This list contains the 118 elements of chemistry. Magnesium Mg 12 24.305 Zinc Zn 30 65.39 Manganese Mn 25 54.938 Zirconium Zr 40 91.224 Molar masses based on 12 C 12.000 amu Numbers in parentheses give the mass number of the most stable isotope. click on any elements name for further information on chemical properties, environmental data or health effects. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). Periodic table Elements molar mass Chemical elements listed by atomic mass The elemenents of the periodic table sorted by atomic mass. Therefore, the unknown gas is most likely argon. 
C Add together the masses to give the molecular mass. Therefore, the unknown gas is most likely argon. B Obtain the atomic masses of each element from the periodic table and multiply the atomic mass of each element by the number of atoms of that element. Examination of the periodic table reveals that Argon has a mass of 39.948 g/mol. Use this periodic table for calculating molar mass for any chemical formula. For radioactive elements with no stable isotopes, mass number of the most stable isotope is shown in brackets. This list contains the 118 elements of chemistry. B The calculated molar mass is 33.9 g/mol. Conventional atomic weights are used for boron, bromine, carbon, chlorine, hydrogen, lithium, magnesium, nitrogen, oxygen, silicon, sulfur and thallium. 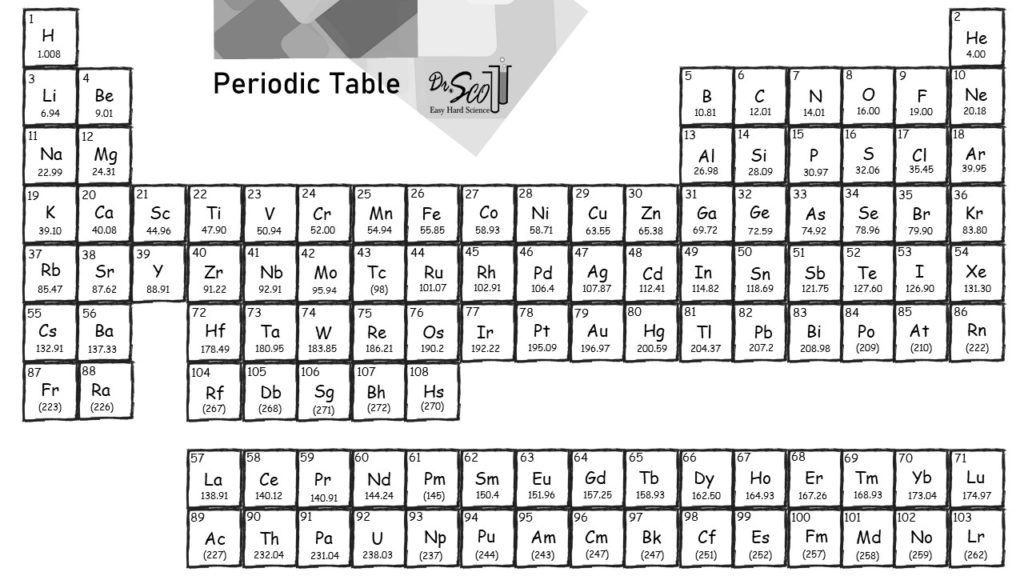
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
